PFAS Leachate Treatment Breaking Down the Bond Barrier – Part 1: Treatment Basics
May 20, 2021

The solid waste industry has had well-established methods to treat leachate on-site at landfills or off-site at wastewater treatment plants for decades. Despite some of the complex challenges with leachate characteristics, engineers have known how to remove organic and inorganic contaminants and have done so with efficiencies necessary to meet evolving regulatory requirements.
So why it is so difficult to remove per- and polyfluoroalkyl substances (PFAS) from leachate? Why are these compounds so different from other parameters we have known how to treat for decades?
The same properties that make PFAS such a desired additive to so many consumer and industrial products and manufacturing processes are the same properties that have resulted in the increase in research and regulatory attention over the last few years. The carbon-fluorine bond is the primary reason that conventional treatment methods used to treat the organics and inorganics typically found in leachate are not sufficient to breakdown PFAS.
The treatment of PFAS in leachate has been a topic of discussion within the solid waste industry for the last few years. There have been numerous presentations and articles that touch on currently available technologies, or “of-the-shelf” options, as well as innovative technologies to treat PFAS in leachate. Until a few years ago, the “off-the-shelf” technologies were probably never thought to be used for treatment of these “forever compounds.” Thanks to the affinity of PFAS for these technologies, they are being adapted for removal of PFAS from leachate and other liquid waste streams.
Why is the science behind PFAS compounds and the treatment technologies so important? If the goal is to remove these ubiquitous compounds from the environment, we need to fully understand the implications of these treatment options relative to the unique properties of these compounds.
Currently, technologies used to treat PFAS in leachate fall into 3 categories: destruction, sorption and separation.
Destruction technologies aim to break down the PFAS compound, but the extent in which this occurs will vary depending on the type of treatment. Thermal destruction is typically a more complete approach since it occurs at high temperatures. Although additional research is needed to understand the efficiency of the thermal destruction of PFAS compounds, it is hypothesized that under specified conditions (e.g., time, temperature, and turbulence) the PFAS molecule may be completely destroyed.
There are other options beyond the use of high temperatures which would be considered non-thermal methods. The use of non-thermal degradation (e.g., oxidation methods) could result in an incomplete conversion of the PFAS compound and generation of non-PFAS compounds. The reason for this uncertainty is due to the currently available science on whether or not the destruction of PFAS is complete or partial. What does it mean for a PFAS compound to be completely destructed? Complete destruction involves breaking chemical bonds such that only elemental compounds exist (e.g., CO2, F, etc.) as shown below. To do this, typically a thermal process (such as incineration) is considered, but other techniques (e.g., electrochemical oxidation, photolysis couples with a catalyst) may hold promise. Thermal treatment of PFAS is recommended as the preferred method for the treatment of PFAS based on the interim EPA guidance. However, the extent to which thermal treatment strategies, such as using gas flare technologies or waste-to-energy facilities, provides complete destruction has not been fully evaluated.
Complete Destruction
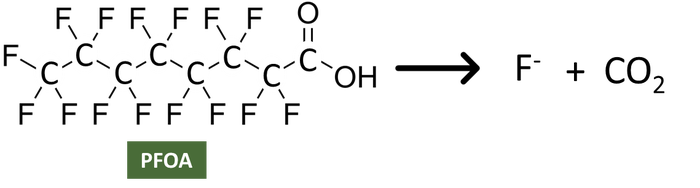
If only partial destruction is achieved then you would expect to still have PFAS compounds remaining, but those could include the initial compounds being treated and then some derivative (i.e., a new compound). This uncertainty is a result of the strength of the carbon-fluorine bond and the possibility that the treatment method only partially breaks the bonds, which results in more a transformation of a PFAS compound. Therefore, when thinking about total PFAS concentrations, if only partial destruction occurs this method would still leave PFAS to be managed, but transforms the compounds into a different mixture to manage which may still contain PFAS.
Partial Destruction
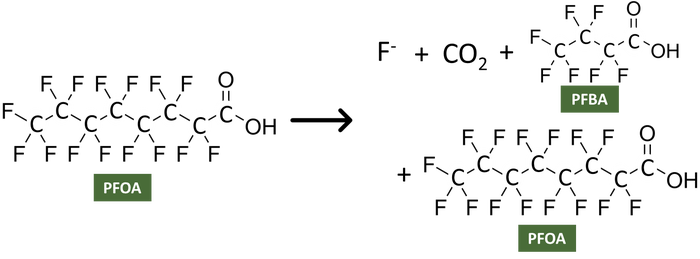
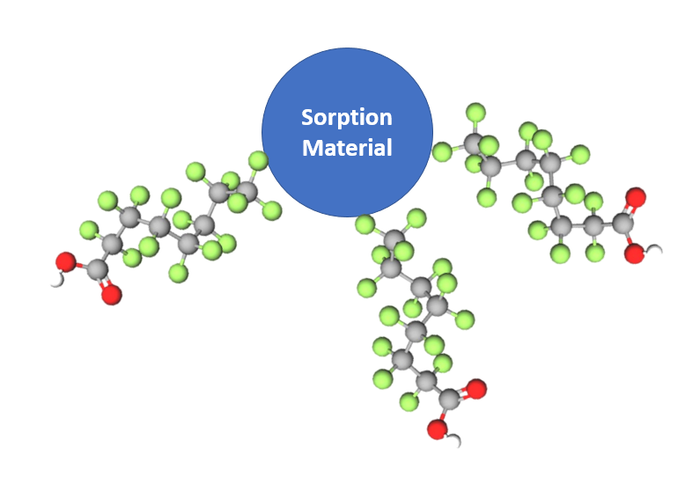
Sorption mechanisms rely on the affinity for PFAS to physically attach to the surface of a solid material in order to remove that compound from the waste stream. When PFAS is removed and sticks to the surface of this material, this mechanism ultimately concentrates PFAS, which generates a more concentrated waste stream that would require additional management and treatment.
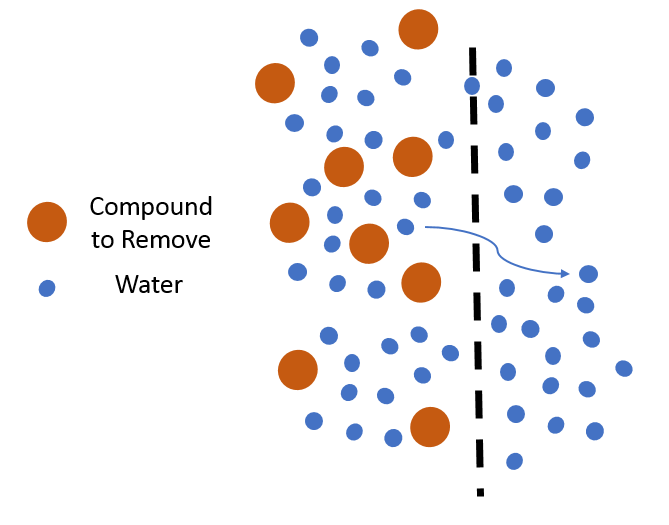
Lastly, treatment can be achieved through separation technologies typically referred to as filtration using membranes. These technologies use the size of the contaminants that should be removed as a method of removal. The approach is to pick a membrane that has holes smaller than the compounds you want to remove so that they cannot pass through the membrane and exit the treatment process. But similar to the sorption approach, a concentrated waste stream containing the compound that is being removed will still need to be managed. One approach that is being proposed to manage the PFAS waste stream, including concentrated streams from sorption and separation technologies is sequestration and solidification. This treatment approach is being evaluated as a means to immobilize PFAS and sequester them in a repository. This approach would essentially remove PFAS from one point of exposure.
Available “off-the-shelf” technologies that have been shown to be effective at managing PFAS in leachate include granular activated carbon (sorption), ion exchange (sorption), and reverse osmosis (separation). The table below highlights attributes of these technologies and their effectiveness at PFAS removal. Data gathered thus far suggest that all technologies remove at least 90% of PFAS from leachate. However, these technologies, since they do not destroy PFAS, result in a concentrated PFAS by-product that must be managed. Therefore, such technologies do not remove PFAS from the environment, but require further steps to be taken to either destroy or sequester the concentrated by-product. Beyond these well-established technologies, there are additional advancements in the field to look at applying other on-the-shelf technologies to remove PFAS from leachate.

About the Author(s)
You May Also Like


