Diagnosing and Understanding Elevated Temperature Landfills (Part 3)
In this article, we describe several chemical reactions that release energy and could contribute to initiating or sustaining an ETLF.
This is Part 3 of a three-part series. Click for Part 1 and Part 2.
A small percentage of municipal solid waste (MSW) landfills in North America have reported elevated and increasing temperatures above those normally associated with a hot gas well. Temperatures above 65 °C (150°F) or even 120 °C (250°F) have been reported in some cases.
In Part 1 of this three-part series on elevated temperature landfills (ETLFs), we described some of the challenges associated with ETLFs. We reviewed the difference between a fire that requires oxygen and subsurface processes in ETLFs that occur in the absence of oxygen, and explained appropriate gas collection system management strategies.
In Part 2, we described biological reactions in landfills, the relationship between these reactions and leachate and gas composition, and how leachate and gas composition can be altered by elevated temperatures.
In this article, we describe several chemical reactions that release energy and could contribute to initiating or sustaining an ETLF. These reactions include (1) metal corrosion, (2) ash hydration, (3) acid-base reactions, and (4) pyrolysis.
With the exception of pyrolysis, the chemical reactions described in this article, as well as the biological reactions described in Part 2, occur in nearly all landfills. We are studying the role of these processes, including pyrolysis, in ETLFs.
Metal Corrosion
Metals that are present in their elemental form (i.e., not combined with other elements) will react with moisture under anaerobic conditions and release hydrogen, in a process described as corrosion. The presence of metals in MSW, particularly aluminum and iron, has decreased over the past 20 years as a result of increased recycling. However, metals persist in MSW and are also present in other wastes that are disposed in landfills, including ash from waste-to-energy facilities, shredder residue, and industrial wastes (e.g., foundry byproducts, aluminum byproducts).
Both iron (Fe) and aluminum (Al) undergo a corrosion reaction under anaerobic conditions:
Fe(s) + 2H2O(l) → Fe(OH)2 + H2(g) ΔH = 47.5 kJ/kg Fe (1)
Al(s) + 3H2O(l) → Al(OH)3 + 1.5 H2(g) ΔH = −15922 kJ/kg Al (2)
where H is hydrogen, O is oxygen, and H = is the energy associated with the reaction (positive requires heat to proceed, negative releases heat). Both reactions oxidize the elemental metal and produce hydrogen (H2). The energy (H) associated with the Fe corrosion reaction is positive, which means that the reaction does not release heat. The aluminum reaction (Reaction 2) produces heat.
Both iron and aluminum reactions produce H2, a highly desirable substrate for methane-producing microbes that produce CH4 and heat by the reaction:
4H2 + CO2 → CH4 + 2H2O ΔH = −31630 kJ/kg H2 (3)
Thus, both iron and aluminum corrosion can contribute to heat generation, either directly through the corrosion process (aluminum) or indirectly through subsequent biological reactions that consume the H2 generated by corrosion. The rate of heat release is directly proportional to the rate of corrosion and the rate of the microbial process. As described in Part 2 of this series, the microbial process can be inhibited by the heat in an ETLF and this is one mechanism by which H2 may accumulate in ETLFs.
Hydration and Carbonation Reactions
Heat released by wetting (i.e., hydration) occurs when water present in a landfill hydrates an oxidized form of various metals (i.e. FeO or Al2O3) or minerals (CaO). For example, calcium oxide (CaO) present in ash is hydrated as in the following the reaction:
CaO + H2O → Ca(OH)2 ΔH = −1164 kJ/kg CaO (4)
The heat generated by hydration is site-specific and waste specific. For example, consider a combustion ash rich in calcium. If the ash is dry when disposed, all of the calcium may be in the form of CaO, with heat released when the ash hydrates in the landfill. However, if ash has been recovered in a wet scrubber process, or water has been added (e.g., to cool the ash prior to disposal), then some of the calcium may be in the form of Ca(OH)2, with the heat of hydration released prior to disposal. Thus, the manner in which waste is handled and disposed at an MSW landfill affects the amount of heat generated and released within the landfill.
Similar to hydration, heat can be released by reactions that use CO2. These are referred to as carbonation reactions that proceed when certain chemical components (e.g. OH or Cl) are replaced by gaseous CO2. Carbonation reactions release an amount of energy similar to hydration reactions. Therefore, if an oxidized form of a mineral is disposed in a landfill, it can release heat through two sequential reactions; hydration followed by carbonation. Reaction 5 is an example of a typical carbonation process.
Ca(OH)2 + CO2 → CaCO3 + H2O ΔH = −1520 kJ/kg Ca(OH)2 (5)
Carbonation reactions are inevitable in landfills given the abundance of CO2 available.
Acid-Base Neutralization
Acid-base neutralization reactions that occur in landfills release energy. The most relevant scenario is neutralization of fermentation intermediates that accumulate as volatile fatty acids (VFAs) (propionate, butyrate, and acetate, see Part 2). VFAs accumulate when microbial processes that produce and consume these acids occur at different rates. If the accumulating acids migrate to a section of a landfill in which methanogenic processes are working in a typical manner (e.g., migrating in leachate percolating through the waste), the acid will be neutralized by active methanogenesis and heat will be released. For example, if all carboxylic acids are present as acetic acid (CH3COOH), then the following neutralization reaction will occur:
CH3COOH + OH− → CH3COO−+ H2O ΔH = −945 kJ/kg CH3COOH (6)
The acid neutralization potential provided by the hydroxide ion (OH-) in Reaction 6 could come from lime [Ca(OH)2], calcium carbonate (CaCO3), or other compounds in the waste.
Pyrolysis
Pyrolysis is the thermochemical decomposition of organic matter at elevated temperature in the absence of oxygen. Anaerobic pyrolysis reactions are distinct from combustion reactions associated with a landfill fire. Part 1 of this series described the difference between indicators of a landfill fire and indicators of exothermic subsurface reactions in ETLFs.
Many sources of organic matter in landfills can be involved in pyrolysis reactions, including paper, yard waste, and wood, which contribute cellulose, hemicellulose, and lignin, as well as food waste, which contributes starches, fats, and proteins. Other types of waste (e.g., plastics, rubber, synthetic textiles) may also contribute to pyrolysis reactions.
Pyrolysis produces gases (CO2, CO, H2, CH4, other volatiles) along with liquids and tars (polyaromatic hydrocarbons and oxygenated hydrocarbons) and leaves a solid residue enriched in carbon, termed “char”. The end products are affected by waste composition, moisture content, pressure and temperature. Typical volatiles include ethane, propane, and hexane while acetaldehyde, propionate and phenols are typical oxygenated hydrocarbons. Factors suspected of influencing pyrolysis include accumulation of volatile gases that can function as reactants, leading to reactions that can become self-sustaining.
Charcoal production from biomass by pyrolysis with the addition of heat and pressure is an analog to pyrolysis in landfills. Studies of commercial charcoal production have shown that pyrolysis energetics transition from endothermic to exothermic in the presence of stagnant gas adjacent to reacting solids combined with elevated pressure (Figure 1). The applicability of the charcoal production analogy to pyrolysis in landfills is a topic of our ongoing research. However, one implication for landfills is that pyrolytic reactions in waste likely are influenced by gas pressure and gas-phase reactants. Thus, we believe that actively managing landfill gas at ELTFs is important.
Discussion
There are a number of chemical reactions that contribute heat to the waste in landfills, including metal corrosion, hydration and carbonation reactions, and acid-base neutralization. These reactions are likely to occur in most landfills, but the reaction rates and the associated heat release rates within a landfill are not well understood.
By themselves, these reactions probably do not provide sufficient energy to be directly responsible for the widespread elevated temperatures encountered in ETLFs. We hypothesize that heat may accumulate in a localized area within a landfill as a result of one or a combination of the microbial and/or chemical reactions described in this three-part series. This localized heat accumulation, when combined with insufficient gas removal and possibly elevated pressure, is hypothesized to promote a transition from endothermic to exothermic pyrolysis, which then becomes self-propagating. A complicating factor is that pyrolysis in landfills is poorly understood, including if and/or when the reaction consumes energy (endothermic) or releases energy (exothermic). We believe that understanding the energetics of pyrolysis is critical to describing the mechanisms by which heat self-propagates in an ETLF. We are currently conducting laboratory experiments to understand MSW pyrolysis and the transition from endothermic to exothermic conditions.
Our evaluation of data from several ETLFs indicates that the reactions and heat and mass transport mechanisms are site-specific and interrelated, making them complex. This suggests that simple interpretations and explanations are not likely to fully describe the initiation and evolution of an ETLF, or provide the mechanistic principles needed to design remedies. Through our research, we intend to understand how the interrelated processes within landfills combine to initiate an ETLF, how these processes are sustained, and what strategies are most effective in preventing and managing ETLFs.
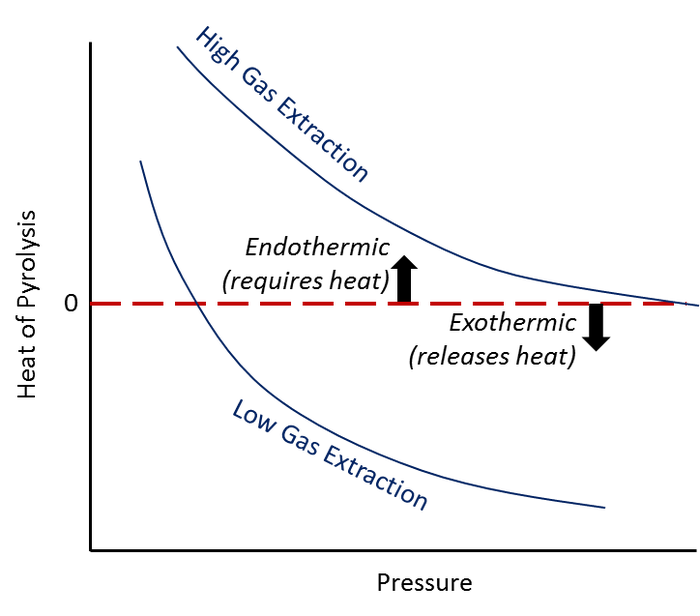
. Influence of Pressure and Gas Extraction on the Energetics of Biomass Pyrolysis for Charcoal Production (adapted from The Art, Science, and Technology of Charcoal Production by Antal and Gronli).
Figure 1. Influence of Pressure and Gas Extraction on the Energetics of Biomass Pyrolysis for Charcoal Production (adapted from The Art, Science, and Technology of Charcoal Production by Antal and Gronli).
About the Author(s)
You May Also Like


